Electric vehicles have reached a milestone that once seemed distant and now feels almost inevitable. More than one in ten new cars sold globally is electric. This shift has delighted policymakers and unsettled incumbents, but it has also given rise to a new anxiety, one that spreads quickly online. If millions of cars run on large batteries, what happens when those batteries wear out?
The question is often posed with quiet certainty, as if the answer were already known and not especially encouraging. A popular line of argument suggests that electric vehicles merely trade tailpipe emissions for a future glut of toxic waste. Social media, that great amplifier of half-truths, has turned this into a recurring debate.
A typical exchange captures the overall sentiment. One electric vehicle owner reports a colleague’s concern that the battery in a Tesla electric vehicle will eventually become an environmental liability. Others respond with equal confidence that recycling is already viable and even profitable. A third group points out that many batteries do not head straight to recycling at all. Instead, they are repurposed for stationary storage, powering homes or supporting solar installations long after they have been retired from automotive duty.
Each of these claims contains a sliver of truth. Taken together, they reveal less about batteries than about the fragmented way in which people understand them.
The industrial reality is more prosaic and, in its own way, more reassuring. Modern processes can recover more than 90% of the valuable metals contained in lithium-ion batteries. That figure alone undermines the notion of an impending waste crisis. More importantly, the timing is often misunderstood. The bulk of electric vehicles on the road today are relatively young. Their batteries, designed to last well over a decade, are not about to flood recycling facilities. The much-feared wave of end-of-life batteries remains several years away.
This delay is not a problem. It is an opportunity.
Recycling infrastructure, like any other form of heavy industry, benefits from time. Processes can be refined, plants can be scaled, and supply chains can be organized before volumes surge. The electric vehicle transition, for all its speed, still offers a window for preparation.
The case for recycling extends beyond environmental tidiness. It is also about economics, energy efficiency, and geopolitics. Metals such as copper and aluminum have long been recycled at high rates, not out of altruism but because it makes economic sense. Recycled aluminum, for instance, requires a fraction of the energy needed to produce it from raw ore. The same logic applies to battery materials.
Lithium-ion batteries contain a cocktail of valuable elements, including lithium, cobalt, nickel, and manganese. Extracting these from the earth is costly, energy-intensive, and often geopolitically fraught. Recovering them from used batteries offers a more stable and potentially cheaper alternative. For countries with limited domestic mineral resources, this is not merely convenient. It is strategic.
Yet batteries are not aluminum cans. Their complexity complicates matters.
Unlike bulk metals, battery materials are embedded in intricate chemical structures. Extracting them requires more than melting and reshaping. It demands processes that can separate, purify, and reconstitute materials without degrading their usefulness. Two broad approaches dominate the industry.
The first, pyrometallurgy, is the older and more established method. It involves smelting shredded batteries at high temperatures, recovering metals such as cobalt, nickel, and copper. The process is robust and scalable, qualities that engineers tend to admire. It is also blunt. Lighter elements, including lithium and aluminum, are often lost in the process. Energy consumption is high, and emissions must be carefully managed.
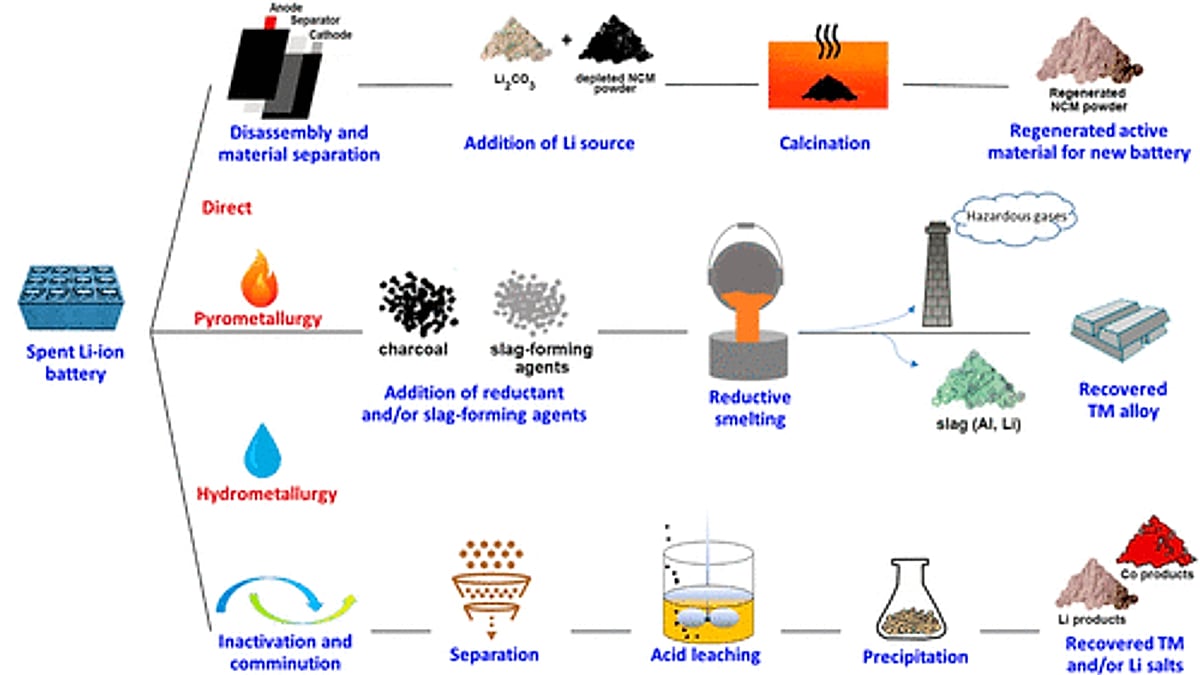
The second approach, hydrometallurgy, is more delicate. Batteries are first discharged, dismantled, and shredded into what is known as “black mass.” This material is then dissolved in chemical solutions, allowing individual metals to be extracted through a series of controlled reactions. Techniques such as solvent extraction, ion exchange, and pH adjustment are used to isolate specific elements.

The rewards for this precision are considerable. Hydrometallurgical processes can recover nearly all valuable materials, including lithium, with significantly lower energy use than smelting. The trade-off lies in complexity. These systems are slower, more capital-intensive, and reliant on careful handling of chemical waste.
In practice, the industry is converging on a combination of both methods, balancing efficiency with completeness. The choice is less ideological than economic.
Economics, as ever, are decisive.
The profitability of battery recycling depends heavily on what is inside the battery. Nickel-rich chemistries, such as nickel manganese cobalt (NMC), yield materials with substantial market value. A ton of processed battery material can contain thousands of dollars’ worth of cobalt and nickel alone. Lithium adds further value, though typically less.
Not all batteries are so generous. Lithium iron phosphate (LFP) batteries, increasingly popular for their lower cost and improved safety, contain neither cobalt nor nickel. From a recycler’s perspective, this makes them less attractive. The irony is difficult to miss. The very chemistries that make electric vehicles cheaper may also make their recycling less lucrative.
This does not render LFP batteries unrecyclable. It does, however, shift the economic equation. Recycling such batteries may rely more on regulatory support or on the long-term value of recovered lithium than on immediate profits.
Technology may yet alter this balance. Emerging designs, including solid-state batteries, promise simpler material compositions and potentially higher-value components. If realized at scale, they could make future recycling both easier and more profitable. For now, they remain largely confined to laboratories and pilot projects.
While chemists refine processes, policymakers are sharpening rules.
In Europe, new regulations set to take effect in 2026 will introduce “battery passports,” digital records that track a battery’s composition, usage, and lifecycle. The aim is to ensure accountability from production to disposal. Such measures may sound bureaucratic, but they serve a clear purpose. By standardizing information and enforcing recovery targets, they reduce uncertainty and encourage investment.
The implications extend beyond environmental policy. Recycling reduces dependence on imported raw materials, a concern that has grown more acute as supply chains have come under strain. If recycled materials can meet a meaningful share of demand, the strategic landscape shifts. Estimates suggest that effective recycling could cut the need for newly mined materials by as much as 30%.
Industry, for its part, is not waiting for mandates.
In Germany, Duesenfeld GmbH has developed a process designed to maximize recovery while minimizing emissions. Batteries are discharged, dismantled, and shredded under inert conditions to prevent fires. The resulting material is carefully treated to extract metals, electrolytes, and other components, many of which are returned to battery-grade quality. The company reports recovery rates exceeding 90% for key materials, with minimal carbon emissions.
Across the Atlantic, Redwood Materials is building an ecosystem that spans collection, processing, and reintegration into new batteries. Its partnerships with major automakers suggest that recycling is becoming embedded in the industry’s supply chain rather than treated as an afterthought.
Li-Cycle has adopted a “hub and spoke” model, decentralizing collection while centralizing processing. This approach reduces transportation costs and allows the company to scale efficiently. Meanwhile, Ascend Elements is focusing on producing high-quality cathode materials from recycled inputs, effectively closing the loop.
These companies are not merely experimenting. They are operating at industrial scale, processing thousands of tons of material and preparing for much larger volumes to come.
The broader picture is one of cautious optimism. Battery recycling is not a panacea, nor is it a looming catastrophe. It is an evolving industry, shaped by the interplay of chemistry, economics, and policy.
The social media debate, with its confident assertions and dire warnings, reflects a natural discomfort with new technologies. Electric vehicles, by removing the familiar mechanics of combustion, invite scrutiny of what replaces them. Batteries, large and chemically complex, are an obvious target.
Yet the evidence suggests a more measured conclusion. The tools to recycle batteries already exist and are improving. The economic incentives, while uneven, are real. The policy framework is tightening. And crucially, time remains on the industry’s side.
If there is a lesson in all this, it is a modest one. Technological transitions rarely solve one problem without creating another. The success of electric vehicles will depend not only on how they are built and used, but also on how they are dismantled and reused.
In that sense, the battery is not the end of the story. It is the beginning of another cycle, one that is only just getting underway.
What Do You Think?
Do you believe EV battery recycling will actually scale before millions of batteries reach end of life, or are we being overly optimistic?
If you owned an EV, would you prefer your battery to be recycled or repurposed for home energy storage?
Chris Johnston is the author of SAE’s comprehensive book on electric vehicles, “The Arrival of The Electric Car.” His coverage on Torque News focuses on electric vehicles. Chris has decades of product management experience in telematics, mobile computing, and wireless communications. Chris has a B.S. in electrical engineering from Purdue University and an MBA. He lives in Seattle. When not working, Chris enjoys restoring classic wooden boats, open water swimming, cycling and flying (as a private pilot). You can connect with Chris on LinkedIn and follow his work on X at ChrisJohnstonEV.
Photo credit: Provided by author